Search
- Page Path
- HOME > Search
- [Korean]
- Smelting and Recycling of Vanadium
- Ho-Sang Sohn
- J Powder Mater. 2026;33(1):61-73. Published online February 28, 2026
- DOI: https://doi.org/10.4150/jpm.2026.00010
- 1,215 View
- 20 Download
-
 Abstract
Abstract
 PDF
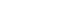
PDF - Global annual production of vanadium is approximately 100,000 tonnes; however, it remains a critical metal for steelmaking and modern industry. This study reviews the current status of vanadium smelting and recycling technologies. Approximately 90% of vanadium is produced as ferrovanadium (FeV) for use in steel alloys, although it is also utilized in titanium alloys, battery materials, and other applications. Both mineral ores and secondary waste resources serve as raw materials for vanadium production. These materials are typically subjected to roasting followed by acid or alkaline leaching to extract V₂O₅. Vanadium metal and FeV are produced from V₂O₅ primarily through aluminothermic reduction; alternatively, metallic vanadium can be manufactured via thermal reduction using Ca, Mg, or C, as well as by molten salt electrolysis. Crude vanadium is subsequently refined into high-purity metal through high-temperature vacuum treatment, electron beam melting, or molten salt electrolytic refining. Vanadium contained in steel scrap is recycled through re-melting in an electric arc furnace. Vanadium present in fly ash and spent catalysts is recovered using smelting processes similar to those applied to natural ores.
- [Korean]
- Synthesis and Investigation of LiVPO4O1-xFxvia Control of the Fluorine Content for Cathode of Lithium-ion Batteries
- Minkyung Kim, Dong-hee Lee, Changyu Yeo, Sooyeon Choi, Chiwon Choi, Hyunmin Yoon
- J Powder Mater. 2023;30(6):516-520. Published online December 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.6.516
- 1,142 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF Highly safe lithium-ion batteries (LIBs) are required for large-scale applications such as electrical vehicles and energy storage systems. A highly stable cathode is essential for the development of safe LIBs. LiFePO4 is one of the most stable cathodes because of its stable structure and strong bonding between P and O. However, it has a lower energy density than lithium transition metal oxides. To investigate the high energy density of phosphate materials, vanadium phosphates were investigated. Vanadium enables multiple redox reactions as well as high redox potentials. LiVPO4O has two redox reactions (V5+/V4+/V3+) but low electrochemical activity. In this study, LiVPO4O is doped with fluorine to improve its electrochemical activity and increase its operational redox potential. With increasing fluorine content in LiVPO4O1-xFx, the local vanadium structure changed as the vanadium oxidation state changed. In addition, the operating potential increased with increasing fluorine content. Thus, it was confirmed that fluorine doping leads to a strong inductive effect and high operating voltage, which helps improve the energy density of the cathode materials.
- [Korean]
- Joint Interface Observation of V and 17-4PH Stainless Steel Dissimilar Materials Manufactured by Direct Energy Deposition
- Se-Hwan Lee, Hobeom Kim, Jeoung Han Kim
- J Powder Mater. 2022;29(1):8-13. Published online February 1, 2022
- DOI: https://doi.org/10.4150/KPMI.2022.29.1.8
- 686 View
- 5 Download
-
 Abstract
Abstract
 PDF
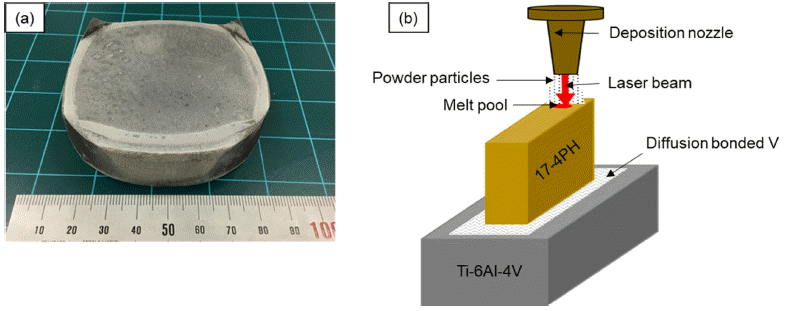
PDF In this study, we have prepared a Ti-6Al-4V/V/17-4 PH composite structure via a direct energy deposition process, and analyzed the interfaces using scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The joint interfaces comprise two zones, one being a mixed zone in which V and 17-4PH are partially mixed and another being a fusion zone in the 17-4PH region which consists of Fe+FeV. It is observed that the power of the laser used in the deposition process affects the thickness of the mixed zone. When a 210 W laser is used, the thickness of the mixed zone is wider than that obtained using a 150 W laser, and the interface resembles a serrated shape. Moreover, irrespective of the laser power used, the expected σ phase is found to be absent in the V/17-4 PH stainless steel joint; however, many VN precipitates are observed.
- [Korean]
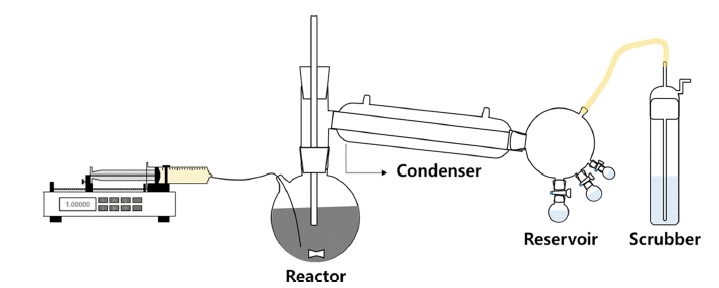
- Study on Manufacture of High Purity TiCl4 and Synthesis of High Purity Ti Powders
- Jieun Lee, Jin-Ho Yoon, Chan Gi Lee
- J Korean Powder Metall Inst. 2019;26(4):282-289. Published online August 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.4.282
- 1,591 View
- 32 Download
-
 Abstract
Abstract
 PDF
PDF Ti has received considerable attention for aerospace, vehicle, and semiconductor industry applications because of its acid-resistant nature, low density, and high mechanical strength. A common precursor used for preparing Ti materials is TiCl4. To prepare high-purity TiCl4, a process based on the removal of VOCl3 has been widely applied. However, VOCl3 removal by distillation and condensation is difficult because of the similar physical properties of TiCl4 and VOCl3. To circumvent this problem, in this study, we have developed a process for VOCl3 removal using Cu powder and mineral oil as purifying agents. The effects of reaction time and temperature, and ratio of purifying agents on the VOCl3 removal efficiency are investigated by chemical and structural measurements. Clear TiCl4 is obtained after the removal of VOCl3. Notably, complete removal of VOCl3 is achieved with 2.0 wt% of mineral oil. Moreover, the refined TiCl4 is used as a precursor for the synthesis of Ti powder. Ti powder is fabricated by a thermal reduction process at 1,100ºC using an H2-Ar gas mixture. The average size of the Ti powder particles is in the range of 1-3 μm.
TOP
 KPMI
KPMI

 First
First Prev
Prev